Top 10 Pharma Solutions for Effective Drug Development?
In today's fast-paced pharmaceutical industry, effective drug development is critical. Numerous challenges arise during the process. Companies seek innovative pharma solutions that address these obstacles. Navigating regulatory requirements is just one example. Each drug must meet rigorous standards before approval.
Pharma solutions provide valuable tools. These include advanced data analytics and clinical trial optimization techniques. However, not every solution is foolproof. Some approaches may lead to unexpected setbacks. Organizations must evaluate their strategies continuously to adapt to changing landscapes. Embracing a flexible mindset is crucial for success.
By exploring the top ten pharma solutions, we can identify what works best. Each solution has its strengths and weaknesses. Understanding these can guide efficient drug development and ensure better patient outcomes. A careful assessment of available resources is essential for informed decision-making. In an evolving sector, improvement is always possible.

Overview of Drug Development Challenges in the Pharmaceutical Industry
The pharmaceutical industry faces significant challenges in drug development. One major hurdle is the high failure rate of clinical trials. According to a report by the Biotechnology Innovation Organization, approximately 90% of drugs that enter clinical trials do not succeed. This statistic highlights the risk involved in the development process.
Regulatory hurdles add another layer of complexity. The average time for a drug to gain approval can take over a decade. In addition, the costs associated with drug development often exceed $2.6 billion. This staggering figure reflects the intense scrutiny and the thorough evaluations required by regulatory agencies. Development teams must balance innovation with compliance, but many fall short of this goal.
Moreover, the landscape of healthcare is continually evolving. Patient needs change, and new diseases emerge. Drug developers must adapt quickly. If they fail to understand these shifts, they risk investing in solutions that don't meet market demands. Continuous research and feedback loops are vital to staying relevant in this competitive arena.
Top 10 Pharma Solutions for Effective Drug Development
| Solution | Description | Benefits | Challenges Addressed |
|---|---|---|---|
| Clinical Trial Management Systems (CTMS) | Software to plan, track, and manage clinical trials. | Improved compliance, real-time data access, better resource allocation. | Streamlining data management, ensuring protocol adherence. |
| Data Analytics Platforms | Tools for analyzing vast datasets to derive insights. | Enhanced decision-making, risk mitigation, quicker identification of trends. | Data overload, lack of actionable insights. |
| Regulatory Compliance Solutions | Systems designed to ensure adherence to regulatory standards. | Lower risk of non-compliance, streamlined submission processes. | Complexity of regulations, evolving guidelines. |
| Collaboration Tools | Platforms that facilitate teamwork across departments and geographies. | Improved communication, faster problem-solving. | Isolation of teams, miscommunication of goals. |
| Artificial Intelligence in Drug Discovery | AI algorithms to identify potential drug candidates. | Reduced time to market, cost savings, enhanced targeting. | High failure rates in initial discovery phases. |
| Patient Engagement Solutions | Tools to enhance patient involvement throughout the trial. | Higher retention rates, more reliable data. | Patient dropouts, lack of feedback. |
| Supply Chain Management Systems | Software for managing and optimizing the drug supply chain. | Improved inventory control, decreased wastage. | Supply disruptions, overstocking. |
| Blockchain for Data Integrity | Using blockchain to ensure data security and integrity. | Enhanced trust in data, improved traceability. | Data tampering, audit complexities. |
| Adaptive Trial Design | Flexibility in trial design to adapt as data comes in. | Efficiency in resource use, maximizing patient outcomes. | Rigid trial designs, lengthy timelines. |
| Biomarker Discovery Tools | Technologies for identifying relevant biomarkers for diseases. | Personalized medicine, improved patient stratification. | Generalized treatment approaches, ineffective therapies. |
Key Phases of Drug Development and Their Importance
Drug development is a complex process with critical phases that shape the outcome of new medications. Each stage, from discovery to regulatory approval, holds unique significance. For instance, the preclinical phase assesses safety and efficacy before human trials. A study by the Tufts Center for the Study of Drug Development highlights that only about 12% of drugs make it through to marketing after clinical trials, emphasizing the need for rigor in early stages.
Clinical trials involve several phases, each designed to answer specific questions about the drug. Phase I focuses on safety, while Phase II examines efficacy. According to the FDA, the average length of a clinical trial can be about six to seven years. This underscores the importance of well-organized trials. Small errors can lead to costly delays or failed drugs.
Tips: Streamline communication between teams. Clarity is essential for success. Regular check-ins can close gaps swiftly. Also, use software tools for tracking progress. They can reveal hidden problems early on. Finally, take time to evaluate feedback. Reflect on what strategies work and which do not. This reflection is vital for continuous improvement in drug development.
Innovative Technologies Revolutionizing Drug Development Processes
Innovative technologies are reshaping drug development. Companies are now leveraging artificial intelligence to streamline research. AI analyzes vast data sets quickly, identifying potential drug candidates more efficiently than human researchers.
Moreover, blockchain technology enhances transparency and traceability. This secures the supply chain and improves data integrity during trials. Additionally, virtual reality offers immersive training for clinical staff, but this approach is still in its infancy. Many researchers express skepticism about its effectiveness.
Bioprinting presents exciting possibilities for personalized medicine, allowing for the creation of tailored therapies. Yet, the technology faces challenges in scalability and cost. The path to full implementation is not straightforward, and further exploration is necessary. The integration of these technologies reveals both promise and areas for improvement.
Top 10 Pharma Solutions for Effective Drug Development
This chart illustrates the effectiveness of various innovative technologies in the drug development process, showcasing their impact on reducing time and costs.
Regulatory Considerations for Effective Drug Development Strategies
Regulatory considerations play a crucial role in drug development. Companies must stay updated on changing regulations. Failing to do so can lead to costly delays. Each stage of development encounters unique requirements. Preclinical studies require extensive data on safety. Clinical trials demand rigorous adherence to protocols.
Submission for approval involves meticulous documentation. Errors can lead to rejection of applications. Companies often underestimate this complexity. Engaging with regulatory bodies early can help. This proactive approach allows identification of potential issues. It may also facilitate smoother approvals later.
The cost of compliance is significant. However, non-compliance carries even higher risks. Small discrepancies can lead to fines or halted studies. Firms need to invest in training staff on regulatory affairs. Keeping a transparent line of communication with regulators is essential. Some companies find this challenging, yet it is vital for success.
Case Studies: Successful Pharma Solutions in Drug Development
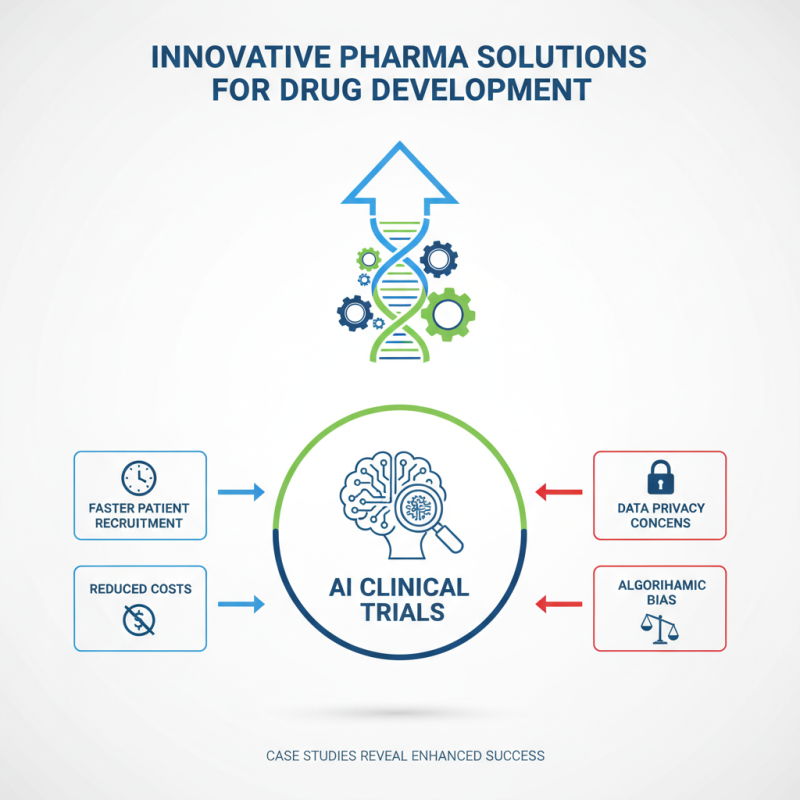
Effective drug development relies heavily on innovative pharma solutions. Case studies reveal various strategies employed by companies to enhance success rates. One notable example involves leveraging artificial intelligence in clinical trials. Companies have used AI to analyze patient data. This approach led to faster patient recruitment and reduced costs. However, it also raised concerns about data privacy and the potential for bias in algorithm outcomes.
Another interesting case is the use of patient-centric approaches. By directly involving patients in the development process, teams gather invaluable feedback. This can lead to better-targeted therapies. However, it can be challenging to manage differing patient opinions. Balancing scientific goals with patient needs requires ongoing dialogue.
Despite these advancements, the path is not without obstacles. Many pharma companies face regulatory hurdles that can slow down development. Additionally, the integration of new technologies can disrupt established workflows. It's crucial for these companies to ensure that all stakeholders are on board. Finding the right balance between innovation and caution is a constant challenge.
Related Posts
-

Top 7 Pharma Solutions Transforming the Pharmaceutical Industry in 2023
-

Unlocking the Secrets of Best Pharmaceutical Manufacturing Technical Specifications and Key Sourcing Strategies
-

10 Essential Insights for Pharmaceutical Manufacturing Success
-

What is Pharma Machinery and How Does It Work?
-

How to Optimize Efficiency in Pharmaceutical Processing Equipment
-

Top 10 Pharma Equipment Trends Revolutionizing Drug Manufacturing in 2023
